 
These results confirmed Rutherford’s atomic theory and provided insights into the inner structure of atoms. The presence of a dense, positively charged nucleus within the atom Most alpha particles passed through the gold foilĪ small percentage of alpha particles were deflected or bounced back 
Rutherford’s Gold Foil Experiment Results Observation The gold foil experiment highlighted the existence of the nucleus and its role in determining the chemical properties of different elements. His findings paved the way for further research and advancements in nuclear physics. 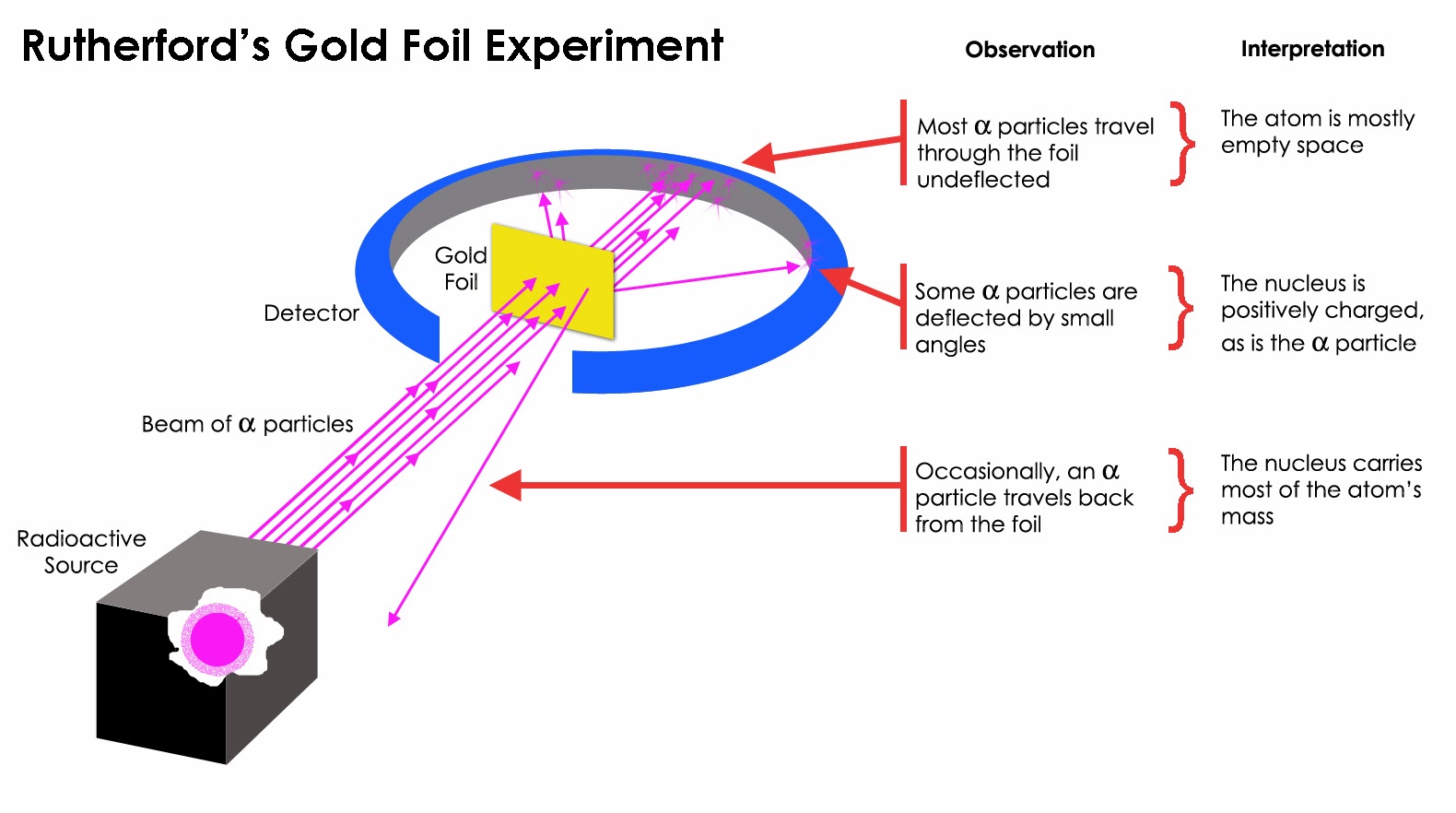
Rutherford’s experiments on atomic structure provided a solid foundation for our understanding of the atom and its constituents. Rutherford proposed that the nucleus is located at the center of the atom, containing the majority of its mass. This discovery challenged the prevailing belief that atoms were indivisible and led to the development of modern atomic theory. However, Rutherford observed that a small percentage of the alpha particles were deflected or bounced back, indicating the presence of a dense, positively charged nucleus within the atom. Most of the particles passed straight through the foil, suggesting that atoms are mostly empty space. In this experiment, a beam of alpha particles was directed at a thin sheet of gold foil. One of his most famous experiments is the gold foil experiment, which provided evidence for his atomic theory. Rutherford’s Experiments on Atomic StructureĮrnest Rutherford conducted groundbreaking experiments on atomic structure that revolutionized our understanding of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
